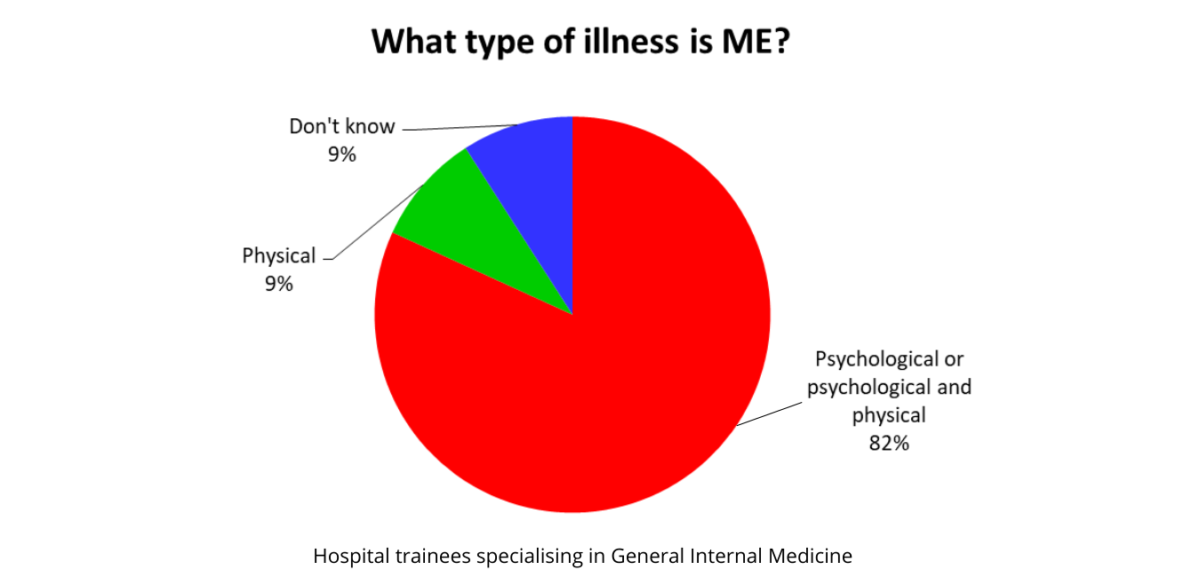
An Audit of UK Hospital Doctors’ Knowledge and Experience of Myalgic Encephalomyelitis Doctors with M.E. Founder and Director Dr. KN Hng, Director Dr. Keith Geraghty and Honorary Fellow Prof. Derek Pheby report their findings regarding ME/CFS knowledge and understanding amongst the medical community following an audit of hospital doctors at a training event in the…Continue readingPoor knowledge of ME/CFS among doctors puts patients at risk of harm
Tag: expertise risk
We are very disappointed to hear of the ‘pause’ in publication of the NICE ME/CFS Guideline, already delayed in April. Following the hard work of the Guideline Development Group, we received news of the further delay of guideline publication with both dismay and profound concern for practitioners, their organisations and patients. Continued delay or deviation…Continue readingRapid Response and Expert Comment: NICE Guideline Delay and Accommodation of Unlawfulness
Care should be taken to reassess existing expertise sources in the field of post-viral disease and to distinguish between 1) the marketability or familiarity of expertise versus 2) disproportionate risk of divergence from legally sustainable standards. It is rare for institutionalised medical norms to exclude scientific consensus to the degree witnessed in this field. This…Continue readingExpertise procurement risk and reputational risk – medical and legal
Although the jurisdictional focus is the United Kingdom, direct equivalence can be found in other jurisdictions and markets. The policy, contractual and legal requirements on medical practice and indemnification provision follow a similar structure in most locales: neither official edict, professional body, private contract, the habitual nature of unlawful care nor eminently misinformed obfuscation can…Continue readingJurisdictions and markets (UK and international)
The absolute clarity of the legal status of ME/CFS is of immediate risk management importance, due its statutory, policy and third-party contractual implications frequently being ignored. These stem from habitually unlawful clinical judgement and arbitrarily discriminative administrative treatment that fail to account for the clarity of classification and associated lawful obligations. The absolute clarity of…Continue readingObligation to not negate ME/CFS legal status
The law does not allow clinical judgement to be discriminatory or to breach duties of care – a normally uncontroversial statement with an unusual degree of unmanaged consequences for this field of disease. The law does not allow clinical judgement to be discriminatory or to breach duties of care – a normally uncontroversial statement with…Continue readingUnlawful clinical judgement (flu and covid vaccination examples)
Informed by the scientific, statutory and policy contexts outlined above, the following simple tests of clinical judgement lawfulness further outline lawful policy implementation requirements, mitigating the elevated probability of normalised unlawful clinical judgement in this context. Contrary conclusions constitute unlawful discriminatory implementation of policy, an arbitrary evasion of clear wording, thus discriminating on the very…Continue readingTests of clinical judgement lawfulness
Neither obfuscation, incorrect pronouncement nor under-informed lack of clarity from governmental, regulatory/self-regulatory or other authorities can change the law, terminal frontline legal requirements nor resultant liability profiles. All decision-making is unavoidably limited by requirements of law, with the elevated likelihood of unlawful decision-making in this field similarly prejudicing top-down communication. The structural difference is only…Continue readingIrrelevance of official edicts and officialised redistribution of liabilities to frontline balance sheets (vaccination and NICE guideline examples)
Unmanaged post-viral disease liability, risk exposure and rising pandemic exposure (draft) Compliance obligations for professionals, operational and expertise risks in practice management, social care, occupational health intermediation, insurance underwriting, claims management and reinsurance Summary The role of habitualised unlawful clinical judgement and discrimination that leads to elevated probability of evidentiable malpractice is outlined. Obligations and…Continue readingCompliance, medico-legal considerations and enforceable obligations
Identically, models cannot excuse discrimination that is substantiated in law. The simple clinical judgement lawfulness tests above apply to the lawfulness of model driven decision-making. Replacing the word “clinician” with “model” in the tests arrives at identical conclusions: a priori assumption of ineligibility/insufficient eligibility of ME/CFS patient cohorts or individuals is unlawful. Identically, models cannot…Continue readingTests of model-dictated eligibility lawfulness (QCovid score example)